Coronavirus (Covid-19)

A collection of articles and other resources on the Coronavirus (Covid-19) outbreak, including clinical reports, management guidelines, and commentary.
A data survey of 1.8 million people in Nebraska showed that XBB.1.5 vaccine effectiveness was 52% against infection and 67% against hospitalization, despite a decline in circulating XBB.1.5 during data collection.
The antibody that mediates the occurrence of VITT associated with adenoviral-based Covid-19 vaccines is nearly identical to the antibodies produced in response to natural adenoviral infection.
In fully vaccinated patients with risk factors for severe disease and patients (vaccinated or unvaccinated) without such risk factors, nirmatrelvir–ritonavir did not significantly shorten the time to alleviation of Covid-19 symptoms.
Investigators found that persons with resolved persistent symptoms after Covid-19 had objectively measured cognition similar to that in persons with short-duration symptoms, although small deficits remained.
Among more than 11,000 participants, 1.7% had at least one false positive rapid antigen test on the basis of RT-PCR results. Persistent false positives tended to occur in women and in persons with autoimmune abnormalities.
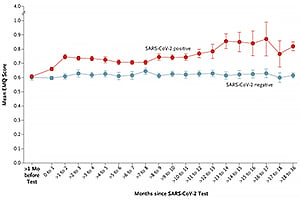
Among Norwegian persons who had prospectively filled out questionnaires evaluating memory, problems with memory were more prominent after positive SARS-CoV-2 tests than after negative tests.
A 36-year-old man was evaluated in the infectious diseases clinic of the hospital because of fevers and throat discomfort that had begun 18 days earlier. An extensive workup was unrevealing. What is the diagnosis?
A 70-year-old woman with advanced HIV infection was evaluated because of cough, shortness of breath, and malaise. Eleven months earlier, she had received a diagnosis of Covid-19. A diagnosis was made.
Simnotrelvir has in vitro activity against SARS-CoV-2. In this phase 2–3 trial in China, simnotrelvir given within 72 hours after symptom onset led to symptom resolution approximately 36 hours faster than placebo.
In this trial of simvastatin involving critically ill patients with Covid-19, which was stopped owing to reduced cases, simvastatin did not meet the prespecified criteria for superiority to control treatment.
A study suggests that prepandemic and SARS-CoV-2–negative cases that are indistinguishable from MIS-C are characterized by Vβ21.3+ T-cell expansion and may be triggered by other pathogens.